
Microbiome Special Issue | PAUL LAWRENCE, BRIANNA SCACCHI, KIMBERLY DEW AND JOSEPH CECCOLI | Biocogent, Stony Brook, New York, USA (P.L., B.S., K.D., J.C.)
Accepted for publication August 2, 2024. Presented at SCC76 and recipient of 2023 Hans Schaeffer Award.
Synopsis
Research into the skin microbiome continues to accelerate at an unrelenting pace with an expanded understanding of how these microbial constituents contribute to skin health. The largest group of these microbes is the bacterial component which includes both beneficial and potentially problematic microorganisms. Some of these exhibit dual roles of commensalism and antagonism with regard to maintaining healthy skin, and as such, skin microbiome modulation efforts should be balanced and precise. To that end, interest has been rekindled in a technology that dates back more than a century, so-called “bacteriophage therapy.” This entails using the natural predators of bacteria in the environment to diminish their population levels viruses (or bacteriophages) that exclusively target bacteria in a species-specific and sometimes strain-specific manner. Here, we present the results of an investigation into using a collection of bacteriophages to target and diminish the levels of Cutibacterium acnes, a member of the skin microbiome that can contribute to the development of acne vulgaris. The bacteriophage cocktail successfully diminished specifically C. acnes both in vitro and in vivo with no adverse effects detected. As other topical microbiome modulatory ingredients exhibit more nonspecific antimicrobial effects on the skin microbiota, this approach offers an attractive, highly targeted alternative.
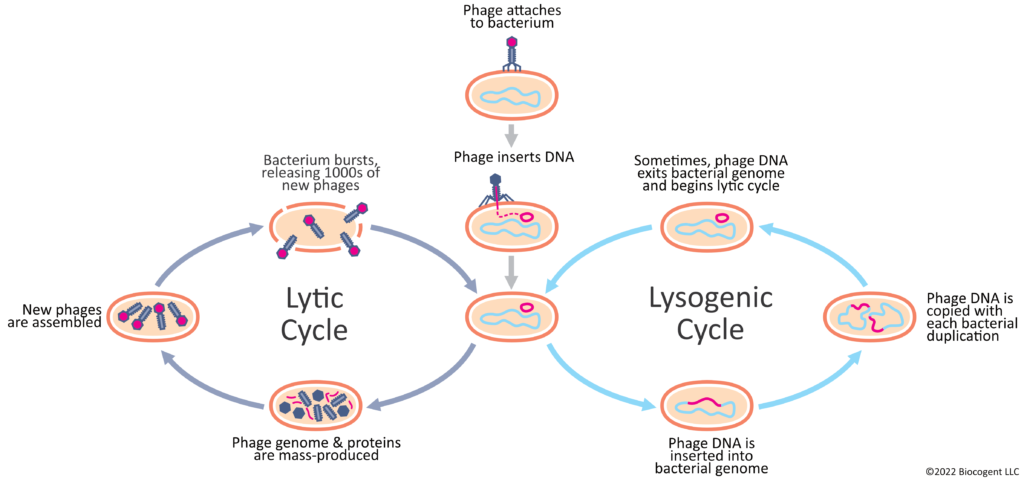
Figure 1. Lytic cycle versus lysogeny. Schematic depicting the differing fates of phages upon infection of their bacterial hosts is presented. They can either actively replicate and assemble new phage particles until the host bacterium is lysed from the inside out (lytic cycle, left side of image), or they can enter into a quiescent state whereby their genetic material lingers and can potentially be incorporated into the host bacterium’s genome (lysogeny, right side of image).
INTRODUCTION
Our understanding of the significant contribution of the skin microbiome to overall skin health rapidly increases with each passing day. Currently, the skin microbiome is one of the most discussed new areas of skin care research—how the various microbial inhabitants of the skin (skin microbiota) contribute to the pathogenesis of many skin diseases and disorders. Indeed, the skin supports a rich community of microorganisms that includes bacteria, fungi, archaea, protists, and viruses. Many of these microbes are considered commensals as their habitation of the skin prevents the encroachment of pathogenic microorganisms. Two of the most abundant bacterial species are the Gram-positives: Cutibacterium acnes and Staphylococcus epidermidis. The former has been established to contribute to the “acid mantle” of the skin that keeps many harmful microbes from getting a foothold through the action of lipases that it secretes. The latter species secretes an enzyme called sphingomyelinase that promotes the production of ceramides that reinforce the skin barrier.
The largest constituent of this vast microbial community are the bacteria, which have been implicated in many skin conditions. The skin bacteriome can vary considerably depending upon which region of the skin is examined giving rise to the study of the “skin biogeography” where specific groups and species of bacteria have been identified that prefer dry, moist, and sebaceous landscapes. For example, S. epidermidis has been found to be abundant in moist regions, while the curiously anaerobic C. acnes exhibits a preference for the oily, sebaceous sites. Indeed, C. acnes establishes colonies deep inside the pilosebaceous units of the skin, where the bacteria construct stratified biofilms and promote sebum production to be insulated from atmospheric oxygen impeding growth. Fluctuations in the homeostatic levels of these various microorganisms are referred to as a “dysbiosis,” and many skin conditions have been attributed to diminished or excessive growth of specific members of the microbiota. One such example is the case of increased growth of Staphylococcus aureus, sometimes at the expense of S. epidermidis, contributing to the incidence of atopic dermatitis. Similarly, although the precise molecular pathogenesis remains to be delineated, and as the name suggests, C. acnes has been suggested to play a key role in the development of acne vulgaris. Acne vulgaris is a fairly widespread skin disease afflicting adolescents and adults from around the world inflicting both physiological and psychological stress on the individual. The classical model of C. acnes involvement in the pathogenesis of blemished skin suggests that higher than normal growth of the bacteria leads to excessive sebum production and induction of various inflammatory pathways.
Related Products


